
I agree Our site saves small pieces of text information (cookies) on your device in order to deliver better content and for statistical purposes. You can disable the usage of cookies by changing the settings of your browser. By browsing our website without changing the browser settings you grant us permission to store that information on your device.
See our Anatomopathology dedicated page to explore all our available tests.
Bleomycin-induced pulmonary fibrosis requires 14 days after a single administration to see significant airway fibrosis as well as associated inflammation. Effects are monitored by whole body plethysmography.
The measurement of various blood metabolites, ions, and enzymes provides a primary screen for function of major metabolic organs such as kidney, liver, gastrointestinal tract, as well as for lipid and glucose homeostasis.
A panel of parameters is proposed, including:
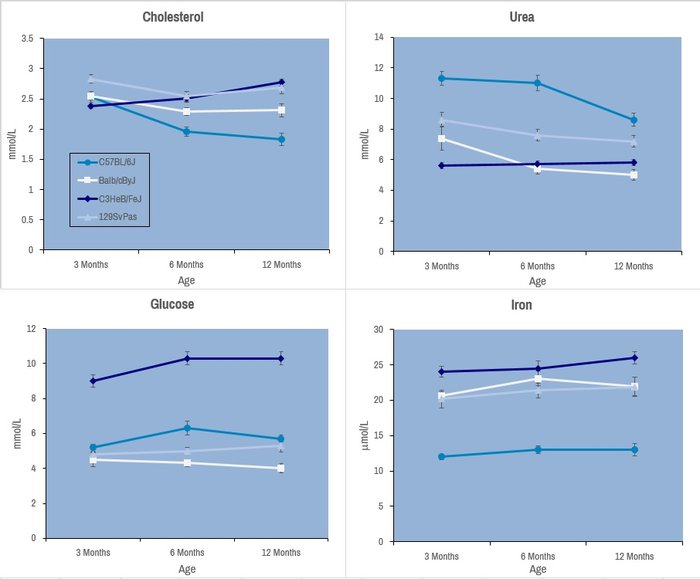
Selected plasma chemistry data from female mice (N=20 per line) of common laboratory strains at different ages (Champy et al. Mammalian Genome 2008). Error bars reflect ± SEM.
These tests are performed with an AU-480 automated laboratory work station (Beckman Coulter France SAS, Villepinte, France).
Exploration of lung inflammatory cells and mediators, e.g. cytokines, contained in bronchoalveolar lavage fluid (BAL). The service includes either the use of standard cytokines and chemokines techniques (Luminex, Elisa) or application of specific protocols provided by users.
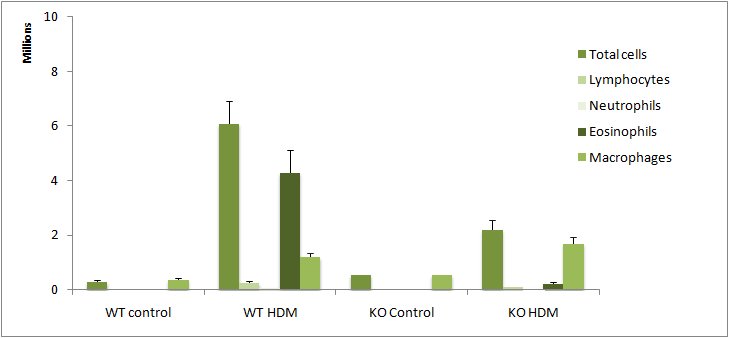
Lung inflammatory cell content in bronchoalveolar lavages (BAL) in House Dust Mite (HDM) model: WT/KO study (C57BL/6 background)
Advia 120 (Siemens AG, Erlangen, Germany)
Cytospin 4
Number of animals: 8 mice/gender/genotype
The intranasal instillation of 3U of elastase is sufficient to establish a meaningful multifocal emphysema in the lungs of mice C57BL6/N after 14 days. This can currently be evaluated by flexiVent™, microCT, and histology.
First-line hematological/immunological analysis of blood or tissue infiltrates for different leukocyte lineages. May be targeted to examine particular immune lineage subclasses.
Exemples:
Thymus:
Spleen:
Blood:
Please see our Immunology dedicated page for more detailed tests.
FACS analysis are performed on a LSR II (BD diagnostics, Le pont de Claix, France).
See our Immunoprofiling dedicated page to explore all our available tests.
Non-invasive tracking of emphysema or tumor growth using computed tomographyic X-ray imaging.
Assessment of airway responsiveness to metacholine-induced brochoconstriction challenge in conscious mice by whole-body barometric plethysmography.
Plethysmography (Penh) allows determination of airway volumes, breath frequency and assessment of airway obstruction in non-restrained mice. This test allows evaluation of fibrosis, as well as physiological and anatomical defects of lungs (e.g. emphysema).
Exploration of airway inflammation and/or airway responsiveness in House Dust Mite-induced lung inflammation.
Advia120
Cytospin 4
Whole-body barometric plethysmograph (EMKA Technologies, Paris, France)
Number of animals - inflammation: 8 mice/gender/genotype
Number of animals - airway responsiveness: 12 mice/gender/genotype
Exploration of airway inflammation and airway responsiveness in LPS-induced inflammation model.
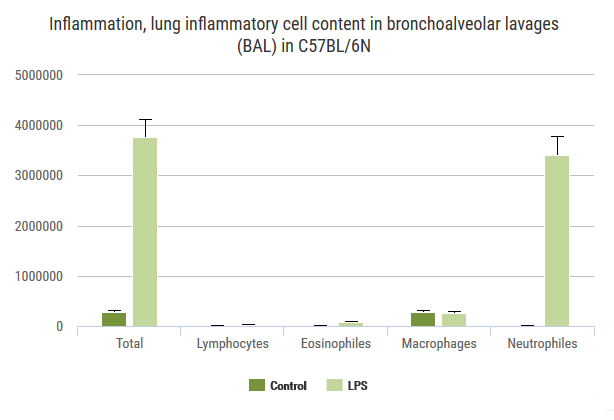
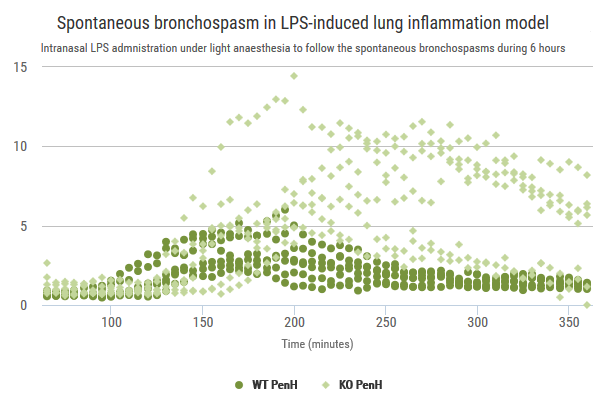
Advia120
Cytospin 4
Whole-body barometric plethysmograph (EMKA Technologies)
Number of animals - inflammation: 8 mice/gender/genotype
Number of animals - airway responsiveness: 12 mice/gender/genotype
We are able to induce eosinophilic inflammation of the lung by presensitizing then exposing the lung to ovalbumin. This can be monitored by whole-body plethysmography, and examined terminally by cytology and cytokine analyses of BALF, immunoglobulin levels in blood and histology.
Use of hypoxia chambers to induce Pulmonary Arterial Hypertension (PAH) for three of four weeks demonstrates significant resemblance to WHO group III categorized PAH, which includes COPD.
Exposure to chronic hypoxia provides a model of COPD and other PAH disorders in mice. For C57BL/6 mice maintained for 21 days at 0% we see highly significant increases in haematocrit, cardiomyocyte surface area, and right ventricular systolic pressure mimicking human PAH.
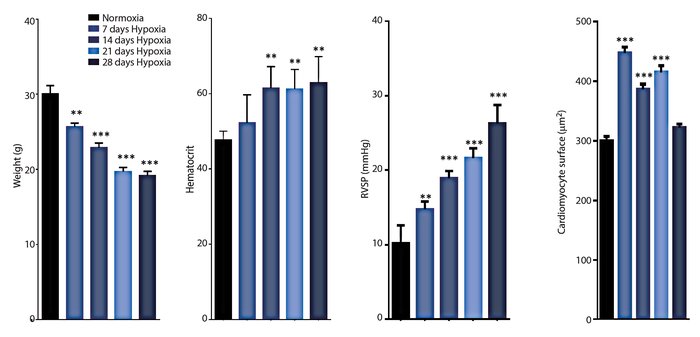
Exposure of C57BL/6N mice to 10% O2 for extended periods resulted in (A) weight loss; (B) increased hematocrit; (C) increase right-ventricular systolic pressure (RSVP); and (D) increased cardiomyocyte surface area.
See our Gene expression analysis dedicated page to explore our different related tests.
See our In vivo Viral Transduction dedicated page to explore our frequently used vectors and delivery modes including stereotactic admininistration (intraventricular and cerebellar)
Controlling infectious diseases requires understanding of the relationships between immune cells and pathogens.
Most current studies to understand infectious processes rely on models requiring
The Biosafety Level 3 Containment Facility (BSL3) has special engineering and containment features that allow investigators to work safely with pathogens classified as risk group 2 or 3. This facility consists of three laboratory suites corresponding to an animal housing room, a laboratory for in vitro experiments and a bio-imaging area.
All instruments are accessible to trained users based on the scientific and regulatory rules established by scientific and biosecurity and safety manager of BSL3. Although the BSL3 platform was only fully operational in May 2015, it already permitted to set up
Real-time imaging studies of immune responses in mice challenged by pathogens such as Salmonella or Brucella infection have led to the development of an immunophenotyping analytical pipeline devoted to infection in synergy with our Immunophenotyping module. The analysis of organ distribution of biomolecules engineered to transport and deliver nucleic acids on living animals using an IVIS Lumina III device is also developed in the frame of a contract with a biotech company. Therefore, we can visualize the spread of infection in vivo, isolate and characterize the pathogens, and analyze phenotypic and functional modification of immune system in the course of infections by advanced live-imaging and flow and mass cytometry.
We provide a broad range of instruments and services for academic and industrial customers. An experienced staff of scientists, technical experts and instrument specialist provides instructions for the design, execution and analysis of standardized multiparametric flow, spectral, mass cytometry and advanced microscopy based studies. All these cutting-edge equipment are technically and operationally enrolled in a regular maintenance program with their commercial providers. Moreover, instrument specialists at PHENOMIN are dedicated to the maintenance and tuning of the instruments on a daily basis. The characterization of the cytometer status and performance using CS&T beads for cytometer set up and tracking system, ensure reproducible performance and instrument sensitivity on a day to day basis. All experiments are standardized using applications setting in order to achieve: (1) high consistency of results over time and (2) facilitate data analysis and results validation. All data are archived and stored on dedicated servers. Depending on the technical complexity and availability, the equipment can be handled by our experts or made available for use to trained customers. PHENOMIN has set up an on line instrument tutorial for training external users. PHENOMIN applies two types of price: one for its academic clients on full cost basis and the other one for its industrial customers taking into consideration intellectual property and the transfer of know-how.
Real-time imaging studies of immune responses in mice challenged by pathogens (Salmonella infection model) and infection-immunophenotyping analysis pipeline in synergy with immunophenotyping CIPHE facility.
Set up vaccine assays and test the ability of adjuvants to enhance vaccine efficacy in a mouse model of infection.
We perform multi-derivation ECG heart in anesthetized mice with 8 probes to generate information about how the electrical activity propagates in 3 dimensions and to determine the electrical axis of the heart. Axis deviations may result from cardiac injury or from a congenital defect.
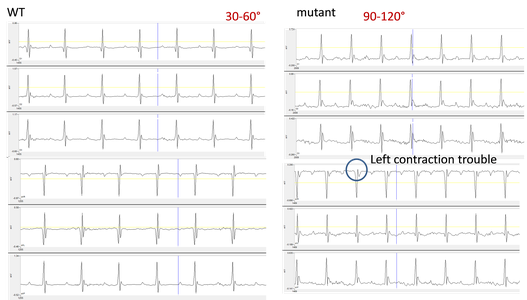
Electrical heart axis difference and specific electrical transmission issues are determined by multiderivational ECG. Multiderivational ECG profiles of a wild-type mouse (left) and mutant mouse (right). Deviated profiles in the mutant animal suggest a left-heart contraction deficiency as well as rotation of the electrical axis to 90-120⁰ from the normal 30-60⁰.
Any strain background can be used
Electrocardiograph ISO DAM8 amplifier (World Precision Instruments, Sarasota, USA) with ECG AUTO v3.3.5.5 (Emka technologies, USA) and 8 lead positions probes.
We recommend 6 mice per group minimum in same anaesthesia conditions and probes positions
Bioluminescence imaging is based on the detection of light emitted as a result of an enzymatic reaction involving a couple “enzyme / substrate”, usually “luciferase / luciferin”. When the enzyme is introduced into systems that are not naturally bioluminescent (cells, bacteria, transgenic animals), these systems play the role of reporter gene and allow to address many biological issues such as gene expression, cell proliferation (cancer) or bacterial dissemination (infectious diseases), for example. A dedicated camera collects emitted photons and the signal is processed to obtain images.
Bioluminescence is performed with IVIS Lumina systems and can be carried out under BSL3 conditions.
Some examples of applications :
IVIS Lumina III (Perkin Elmer)
Fluorescence is based on the excitation of a fluorophore then detection of emitted light. The incident light illuminates the fluorophore with one given wavelength that in return emits photons of longer wavelengths. Thus, fluorescence imaging needs a light source to excite the fluorophore within the targeted tissue, combined with a spectral filter to select the excitation wavelength, a second filter that only allows passage of the emitted fluorescence and finally a detector that detects the signal.
Fluorescence is performed with IVIS Lumina systems as planar epi-fluorescence mainly in the near infra-red wavelengths. Imaging can be performed in vivo (superficial foci) or ex vivo (organs).
Some examples of applications:
Bleomycin-induced pulmonary fibrosis requires 14 days after a single administration to see significant airway fibrosis as well as associated inflammation. Effects are monitored by whole body plethysmography.
The intranasal instillation of 3U of elastase is sufficient to establish a meaningful multifocal emphysema in the lungs of mice C57BL6/N after 14 days. This can currently be evaluated by flexiVent™, microCT, and histology.
Exploration of airway inflammation and/or airway responsiveness in House Dust Mite-induced lung inflammation.
Advia120
Cytospin 4
Whole-body barometric plethysmograph (EMKA Technologies, Paris, France)
Number of animals - inflammation: 8 mice/gender/genotype
Number of animals - airway responsiveness: 12 mice/gender/genotype
Exploration of airway inflammation and airway responsiveness in LPS-induced inflammation model.


Advia120
Cytospin 4
Whole-body barometric plethysmograph (EMKA Technologies)
Number of animals - inflammation: 8 mice/gender/genotype
Number of animals - airway responsiveness: 12 mice/gender/genotype
We are able to induce eosinophilic inflammation of the lung by presensitizing then exposing the lung to ovalbumin. This can be monitored by whole-body plethysmography, and examined terminally by cytology and cytokine analyses of BALF, immunoglobulin levels in blood and histology.
Use of hypoxia chambers to induce Pulmonary Arterial Hypertension (PAH) for three of four weeks demonstrates significant resemblance to WHO group III categorized PAH, which includes COPD.
Exposure to chronic hypoxia provides a model of COPD and other PAH disorders in mice. For C57BL/6 mice maintained for 21 days at 0% we see highly significant increases in haematocrit, cardiomyocyte surface area, and right ventricular systolic pressure mimicking human PAH.

Exposure of C57BL/6N mice to 10% O2 for extended periods resulted in (A) weight loss; (B) increased hematocrit; (C) increase right-ventricular systolic pressure (RSVP); and (D) increased cardiomyocyte surface area.
See our In vivo Viral Transduction dedicated page to explore our frequently used vectors and delivery modes including stereotactic admininistration (intraventricular and cerebellar)